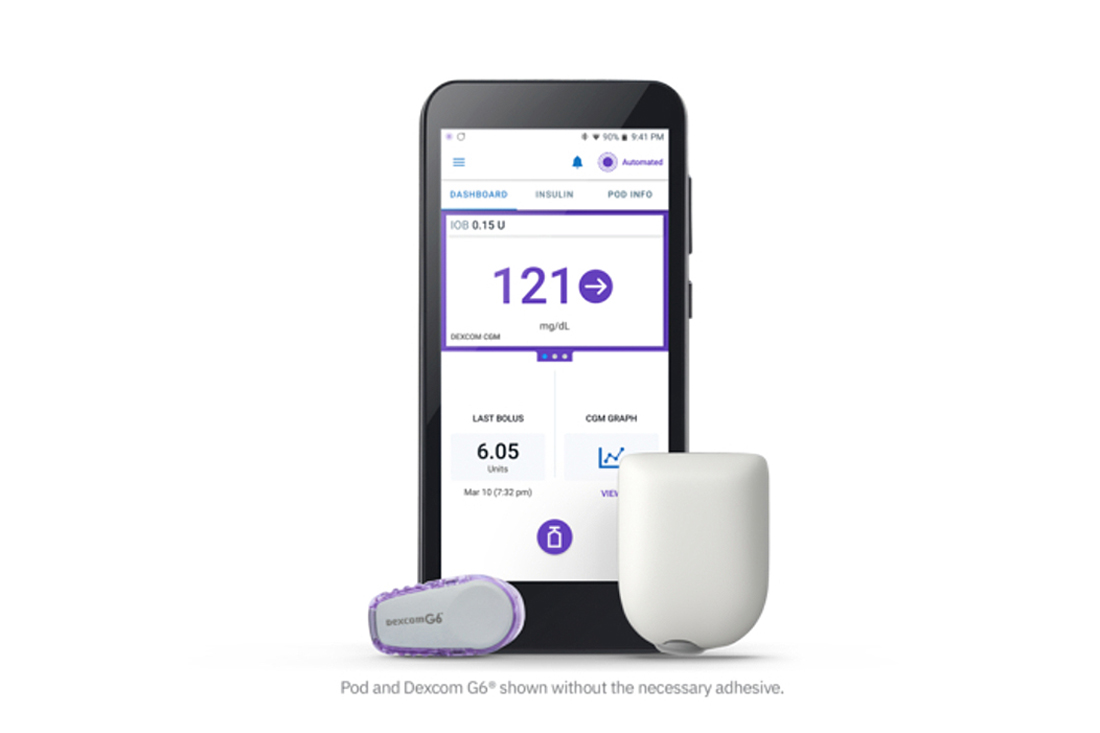
The Omnipod 5 was recently cleared by the FDA after a very long wait!
For those of you folks living in a cave who’ve never heard of the Omnipod, it’s a patch pump without the tubing that’s typically used in traditional pumps, and it can be placed pretty much anywhere on your body. It holds 200 units of fast-acting insulin, and it stays active for 80 hours before it shuts off and a new one needs to be filled and activated. It’s waterproof and you can swim with it (but if you don’t know how to swim, don’t try it just because you got an Omnipod!)
The Omnipod 5 is a hybrid closed-loop AID (automated insulin delivery) device in the same category as the very first Medtronic 670G, the Tandem Control-IQ, and DIY (do it yourself) open source systems such as “Looping”. As someone living with T1D for over 50 years and an endocrinologist specializing in diabetes, I am just amazed how these systems have totally transformed the care of people with type 1, and now we have four systems to choose from!
All of these hybrid closed-loop (HCL) systems with modulating basal rates that deliver insulin based on predicted glucose values from CGMs are designed to improve time in range (the percent of time spent between 70 and 180 mg/dL) while minimizing hypoglycemia. The data presented and published on the clinical trials over the past few years evaluating the Omnipod 5 in adults demonstrate the ability to obtain an A1c very close to 7%, time in range of about 74%, and time below range (lower than 70 mg/dL) of less than 2%. There have been no studies measuring one HCL system against another, but one thing is for certain – these systems perform better in terms of time in range, A1c, hypoglycemia, etc. than pumps and CGM devices that do not communicate with each other! It is so awesome to have choices and to be able to get the system that best fits your individual needs.
So what do you need to get started on the Omnipod 5 system? It’s important to note that the current Omnipod system and Omnipod DASH Pods/PDM are not compatible with the new Omnipod 5 system. The new Omnipod 5 Pod includes a built-in algorithm for automated insulin delivery, Bluetooth for communication with the Dexcom G6 CGM, and the controller software specifically for the Omnipod 5 Pod. Current Omnipod users (those who have DASH or the older Eros Pods) will need to make sure they get the Omnipod 5 Pods and the new controller (which is like an Android phone but without phone capabilities). The new controller will be supplied free of charge. There will be a limited launch of the Omnipod 5 at first, so you may need to be patient. They eventually will be easier to get because they’ll be distributed directly from pharmacies, but you will need a prescription from your doctor. So much better than going through those bureaucratic and obstructive diabetes supply companies!
At this time, only the Android phone can be used as the controller, and not an iPhone. If you are an Android user, you can control your system with your personal smartphone, but if you are an iPhone user you will also need to carry the Android controller.
Some specific features of the Omnipod 5 include:
- The ability to set your goal range between 110 and 150mg/dL
- An exercise glucose mode set at 150 mg/dL with reduced insulin
- Boluses given every five minutes that will increase, decrease, or pause based on your 60-minute predicted glucose
What about compatibility with the Dexcom G7 CGM that may be coming out later this year? Currently the Omnipod 5 is not compatible, but I bet both Dexcom and Insulet (the makers of Omnipod) are working on this issue.
The FDA approval of the Omnipod 5 is awesome news, and it will hopefully increase the number of people with type 1 taking advantage of hybrid closed-loop technology, which will lead to better A1cs, improved time in range with minimal hypoglycemia, reduced acute and chronic complications, and greatly enhanced overall quality of life.
Additional Resources:
TCOYD Podcast #13 – Diabetes & Technology
My Transition from the Medtronic 670G to the Insulet Omnipod/Dexcom Pair
Dexcom’s Next Gen G7 Gets FDA Clearance!

Good information. Thank you. But if your an I phone user you will have to carry one more item in your purse or pocket. Also if you use more than 200 units in 3 days. That might prove tough for insurance.
Yeah I’ve seen that. Some people might need more than 10 per month if they have to replace it sooner.
Well, it’s only approved for Samsung S10 so far. A 3 year old mobile phone that most people never got, or has passed on some time ago. We will see if the app is either approved for newer models soon enough, or “hacked” like the Dexcom App was early on. So we with newer Samsung Android phones (S21 soon S22) could use it too.
Pharmacy distribution is certainly more convenient but Medicare does not normally see insulin pumps as a pharmacy benefit. Currently, even with a PA approved, I’m paying 40% copay = $19/each. My understanding is insulin pumps thru DME usually require 20% copay for Medicare patients. Pharmacy distribution does not appear to be a benefit for Medicare patients.
Agree..such a complicated process!
At one time I thought I read the Omnipod 5 would be compatible with both the Dexcom CGM and the Abbott CGM. I guess that is no longer the case?
The Omnipod 5 is compatible with just the Dexcom G6 only for now. The FreeStyle Libre is on hold for an issue with vitamin C interference with glucose values. The Dexcom G7 application to the FDA was just submitted, so it will be at least 6 months before it gets approved. Perhaps in the future.
Dr Elderman,
So the Dexcom G7 approval will bring the Bluetooth interface between the Omnipod 5 and Dexcom G7 ? I have been waiting for an integrated solution ( like many others) and it seems to be taking forever! I know …. the pandemic. Grrr. Wish FDA would move faster.
The Dexcom G6 isn’t integrated, correct?
The answer to your first question is yes, but that software has to be worked out after the G7 is approved. The Omnipod 5 is all set up to work with the G6 now, and it’ll convert to the G7 once it’s available.
Great news! I cannot wait to get my 5 and get the looping in place – carrying the separate device is well worth what I’d expect to get out of the integration (iPhone user). Thanks for sharing!
Enjoy! and thanks for the post
Totally agree!
I’m curious to know what the cost to run this option will be. 10 pods per month vs. a tubed 670G, which is what I’m using now. The cost is important to me. Also, my warranty expires this mid year and I was considering a tslim. More to debate now. This is a good transition option in the meantime.
OMG. you will love the G5 or the Tslim as it uses the Dexcom CGM with no calibration needed and always in automode…you will be in heaven. We will have more programs on the systems..see the March 19th tcoyd conference
Great review! I’ve been carrying an Omnipod PDM since 2008 so I’m sure I can manage a while longer with the new “hand-held controller” as I hear Insulet folks calling it. Medicare user since 2016 — my DASH pods are covered by Tricare for Life with very low copay. Checking the formulary to see when Omnipod 5 shows up there. Hoping our military T1D families can get access to this new technology soon—since it’s the same price as DASH, it should happen easily. Thanks for all you do!
We hope so too – thanks Sondra!
Currently Looping. Any advantages to switching to the Omnipod 5? Eros pods currently covered as DME with my insurance after 900.00 deductible. Will Omnipod 5 only be covered by pharmacy benefits?
There really are no significant benefits to switching. If you use an iPhone, you’ll have to carry around 2 devices. The algorithm is not as good as the looping algorithm, and lastly, you would not have to carry around the RileyLink with the Omnipod 5. I just found out that pretty soon we will be able to loop with the DASH pods, which means we won’t have to carry the RileyLink.
I am currently using loop with G6 and Eros pods. I will son need to reorder the pods, getting another 4 month supply. I REALLY like the thought PG using Dash pods without the RileyLink and am holding off as long as I can on the Eros reorder.
Any idea the timeframes involved?
Apparently you can get the new pods now.
How do I register for this?
You can sign up to be notified when it becomes available here: https://www.omnipod.com/what-is-omnipod/omnipod-5
Omnipod 5’s algorithm is not as good as the looping algorithm,
What is better about the loop. Very interested in looping with app
Think you just answered your own question :). Give looping a try and see what you think!
actually I forgot to put quotation marks around the statement. That statement was from your website.
I’m still interested why you feel it’s better. I use Omnipod Dash & Dexcom G6. I really want to carry less items with me plus I was pretty confused how loop is set up now with a Riley and Omnipod.
My preference, which may be silly, is to wait for Omnipod 5 and the loop app to be available. iPhone user so I wouldn’t be able to control the pump from my phone which I desperately want.
I appreciate the time you’re taking with me.I do make donations to TCYOD – great information and solutions.
The main reason is that loop is not hampered by FDA rules and regulations, and there are a lot more variables, adjustability and settings you can set yourself vs the Tandem and Omnipod, which is set for you. It’s especially helpful during exercise. It’s very easy to set up an exercise setting that you cannot do with the other two to avoid hypos. You can start looping if you want with the Eros Omnipod, and very soon you’ll be able to loop with the DASH pod, and you won’t have to carry around the RileyLink.
Dr Edelman,
I’ve been watching your videos for the last year on utube and find them incredibly helpful and entertaining!
Are you going to try out Omnipod 5 yourself and let us all know your experience?
I’m upgrading from Dash in a couple of weeks and very excited to test it out myself.
Absolutely, I can’t wait to try it, and I’ve heard great things about it.
I’ve looped with omnipod eros and dexcom G6 for two years. I’m an android user. When I forget my iphone at home, my loop is worthless. One benefit of omnipod 5 is the algorithm resides on the pod. Once the pod is initialized, the loop will adjust without the iPhone (controller).
Yeah, that certainly is one advantage. I’m glad you like it.
I was loving the Omnipod 5 (been using for 6 weeks) until last night. The pod lost communication with my Dexcom and would not reconnect. I followed the on screen directions in the app Tried to reconnect….failed! Deleted and re-entered the transmitter SN….failed! Tried a new pod….failed! Tried another new pod….failed! Called tech support this morning and nothing they’ve told me to do so far has worked. I even went as far as changing pods now for a 4th time and connecting to the controller…also failed.
My CGM Transmitter has connection with the Dexcom App and is working perfectly. I am getting consistent glucose readings. The pod and the Dexcom are just not talking to each other and tech support has no other suggestions other than to leave the house and try to reconnect. Not sure how leaving the house is going to help this situation but I will try.
I waited patiently for for many months for the release of the Omnipod 5 and it has made a significant improvement in my “in range” glucose readings the past 6 weeks,but I am a bit frustrated now that I cannot get it working again. Back to the old way of using my Omnipod until tech support can figure this out. 🙁
If you haven’t already, try tech support again and try to get someone else who is more knowledgeable.
Dr. Edelman
I’m watching your live stream about latest diabetes tech. You described insulin sensitivity factor and basal rates being important to the algorithm working well. While this might be true of the other systems it is not for Omnipod 5. It only takes your programed basal rates into account on pod one. Then it ignores all info you put in and goes off total daily dose history unless you run your pump on manual mode or are manual bolusing.
Thank you, professor!
När kan man räkna med att kunna använda Omnipod 5 ihop med Libre. Har Libre 3 nu och vill inte gärna byta till större Dexcom.
We really don’t know when the FDA will approve the Libre 3 to be used with a pump. My personal feeling is that you should switch to the Dexcom at this point.